 In prokaryotes, the electron transport chain components are found in the plasma membrane.Īs the electrons travel through the chain, they go from a higher to a lower energy level, moving from less electron-hungry to more electron-hungry molecules. In eukaryotes, many copies of these molecules are found in the inner mitochondrial membrane. The electron transport chain is a collection of membrane-embedded proteins and organic molecules, most of them organized into four large complexes labeled I to IV. We will look more closely at both the electron transport chain and chemiosmosis in the sections below. As H + ions flow down their gradient and back into the matrix, they pass through an enzyme called ATP synthase, which harnesses the flow of protons to synthesize ATP. Splitting of oxygen to form water. At the end of the electron transport chain, electrons are transferred to molecular oxygen, which splits in half and takes up H + to form water. This pumping establishes an electrochemical gradient. Some of the energy is used to pump H + ions, moving them out of the matrix and into the intermembrane space. In the process, they turn back into NAD + and FAD, which can be reused in other steps of cellular respiration.Įlectron transfer and proton pumping. As electrons are passed down the chain, they move from a higher to a lower energy level, releasing energy. Reduced electron carriers (NADH and FADH 2) from other steps of cellular respiration transfer their electrons to molecules near the beginning of the transport chain. The key steps of this process, shown in simplified form in the diagram above, include:ĭelivery of electrons by NADH and FADH 2. Together, the electron transport chain and chemiosmosis make up oxidative phosphorylation. Energy released in these reactions is captured as a proton gradient, which is then used to make ATP in a process called chemiosmosis. Electrons are passed from one member of the transport chain to another in a series of redox reactions. The electron transport chain is a series of proteins and organic molecules found in the inner membrane of the mitochondria. The electron transport chain is a series of proteins embedded in the inner mitochondrial membrane. Simple diagram of the electron transport chain. In this article, we will examine oxidative phosphorylation in depth, seeing how it provides most of the ready chemical energy (ATP) used by the cells in your body. Without enough ATP, cells cannot carry out the reactions they need to function, and, after a long enough period of time, may even die. If oxygen is not there to accept electrons (for instance, because a person is not breathing in enough oxygen), the electron transport chain will stop running, and ATP will no longer be produced by chemiosmosis. So, where does oxygen fit into this picture? Oxygen sits at the end of the electron transport chain, where it accepts electrons and picks up protons to form water. In chemiosmosis, the energy stored in the gradient is used to make ATP. In the electron transport chain, electrons are passed from one molecule to another, and energy released in these electron transfers is used to form an electrochemical gradient. Oxidative phosphorylation is made up of two closely connected components: the electron transport chain and chemiosmosis. But have you ever wondered why that is the case, or what exactly your body does with all that oxygen?Īs it turns out, the reason you need o xygen is so your cells can use this molecule during oxidative phosphorylation, the final stage of cellular respiration. As you know if you have ever tried to hold your breath for too long, lack of oxygen can make you feel dizzy or even black out, and prolonged lack of oxygen can even cause death. 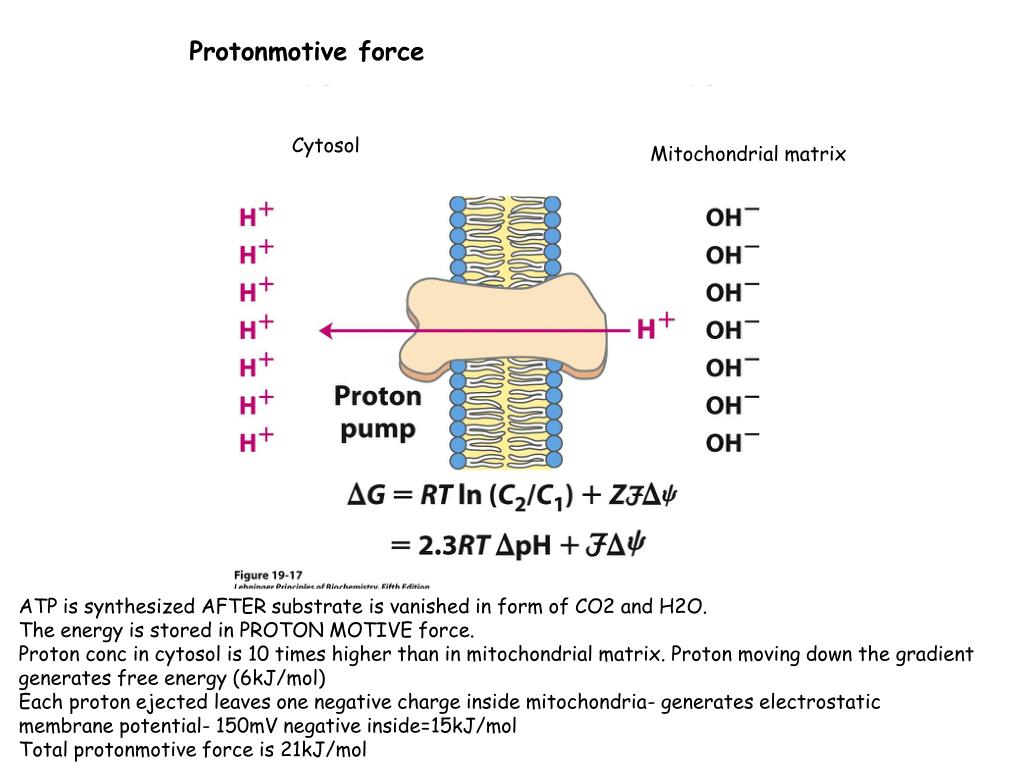
You, like many other organisms, need oxygen to live. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
